The table below gives the number of electrons, protons and neutrons in a substance X, Y and Z. study - Tutorke
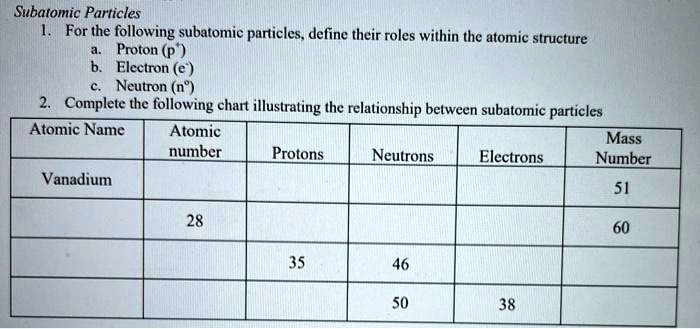
SOLVED: Subatomic Particles For the following subatomic particles. define their roles within the atomic structure Proton (p Electron (e ) Neutron (n") Complete the following chart illustrating the relationship between subatomic particles
I want the symbol,number of protons,number of neutrons,number of electrons,distribution of electrons and valency of the first 30 elements - 3y90wojj

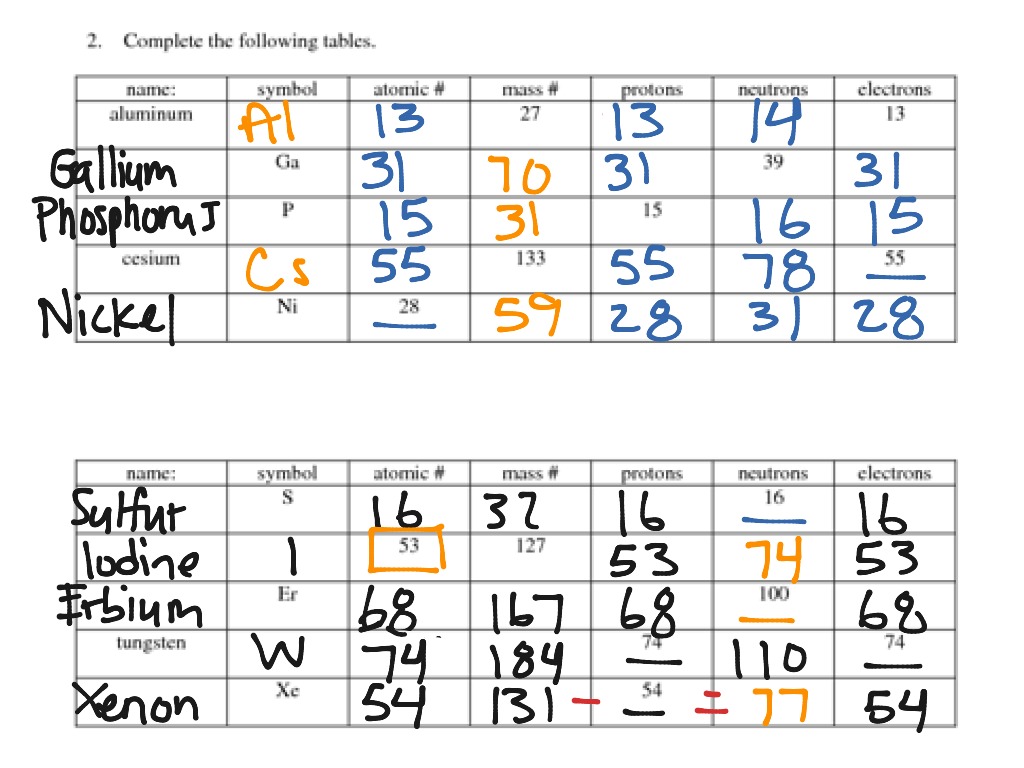
Protons, Neutrons, and Electrons Practice Worksheet | Practices worksheets, Chemistry worksheets, Proton neutron electron

ACROSS1 Maximum number of electrons in L shell5 H^+ ion8 Negatively charged fundamental particles9 Atoms of different elements with different mass numbers, but same number of neutrons10 Atoms of different elements having

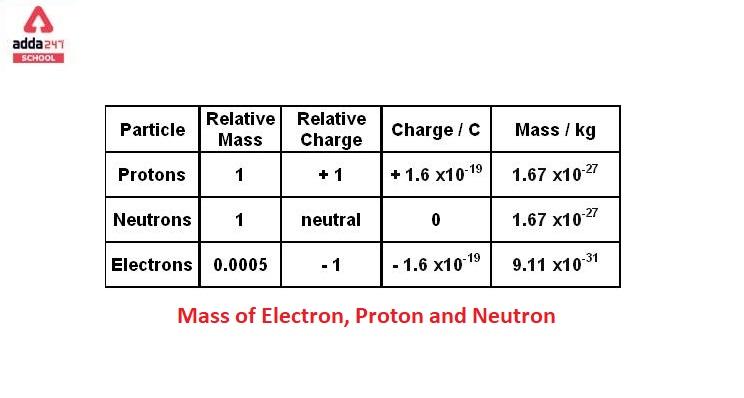



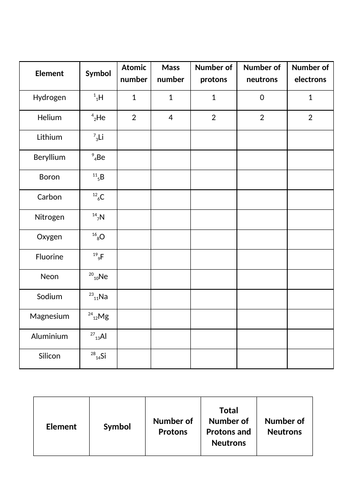
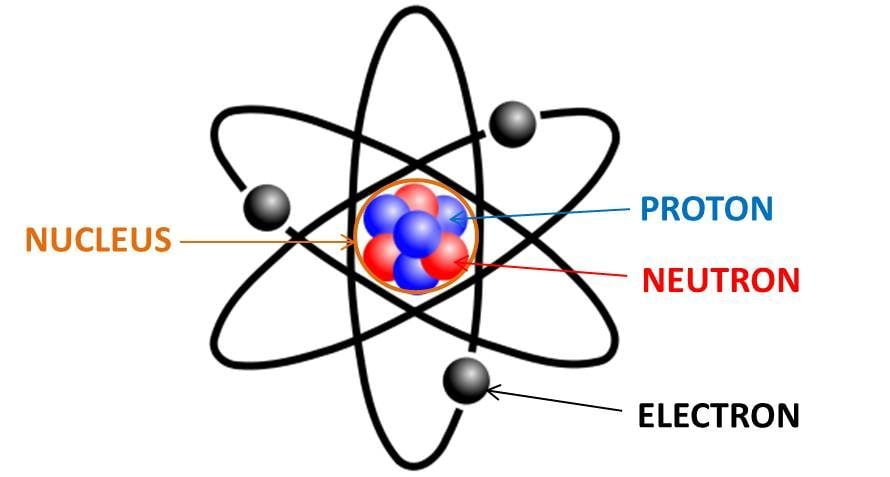


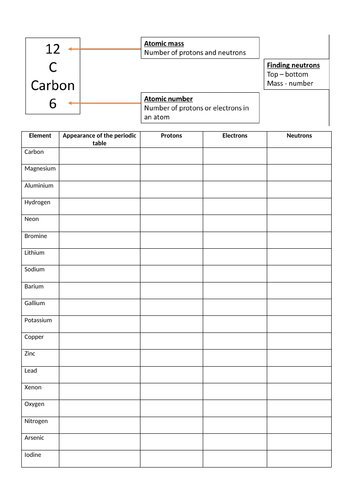
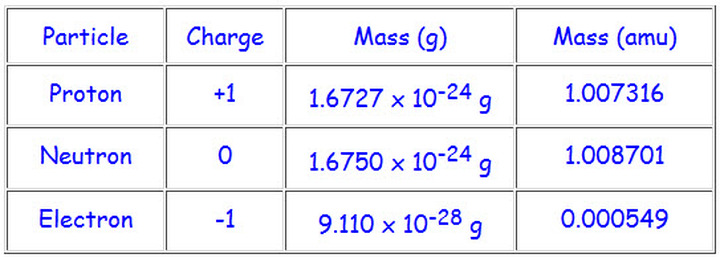
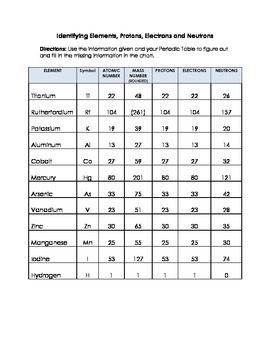

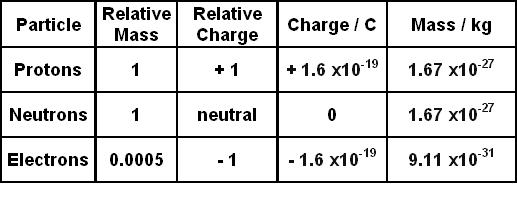
![Malayalam] Symbols of certain isotopes are given in table ,Complete t Malayalam] Symbols of certain isotopes are given in table ,Complete t](https://d10lpgp6xz60nq.cloudfront.net/physics_images/EXP_RF_IX_CHE_P01_C01_E01_032_Q01.png)
